Press Release
Recall of unregistered proprietary Chinese medicines
25 January 2013
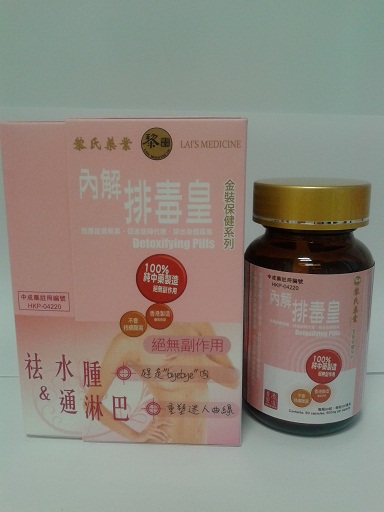
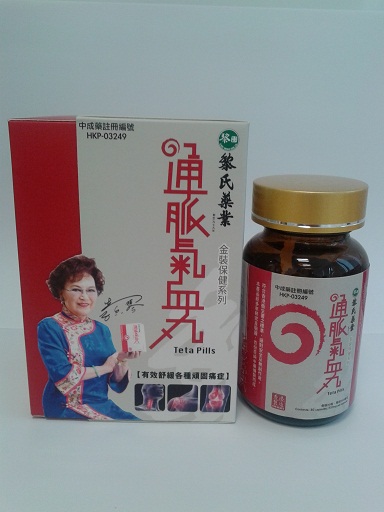
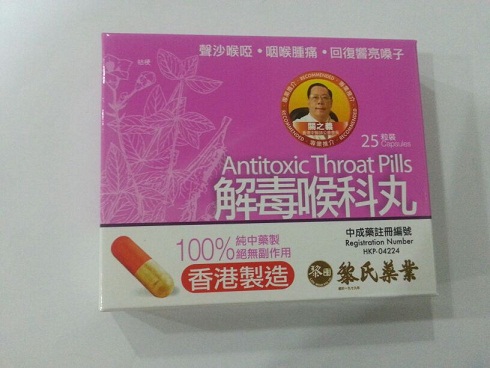
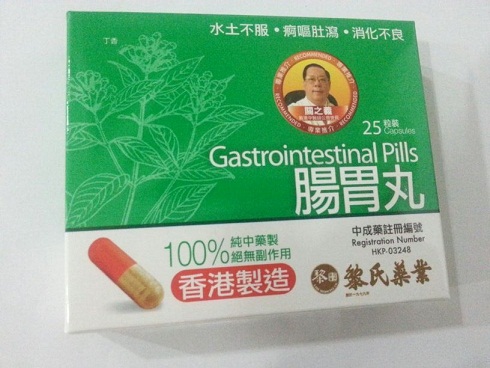
The Department of Health (DH) today (January 25) instructed a licensed manufacturer of proprietary Chinese medicines (pCms), Lai Sing Medicine Factory Limited at 2 Yee Hop Yuen, 11 Miles, Route Twisk, New Territories, to recall from consumers its seven suspected unregistered pCms, namely Teta Pills, Lui Cho Flu & Fever Pills, Cure All Medical Extract, Antitoxic Throat Pills, Gastrointestinal Pills, Detoxifying Pills and Allergic Rhinitis Nose Drops. Details of the seven unregistered pCms for recall are listed in the Annex .
The action is called for after detection of the above products during the DH's routine market surveillance. All of them have not been registered with the Chinese Medicine Council of Hong Kong (CMCHK). Preliminary investigation revealed that the products are manufactured and wholesaled by Lai Sing. Although they share certain similarities with Lai Sing's seven other registered pCms, the pCms concerned differ in product name or dose form.
Lai Sing has set up a hotline at 8102 2208 for related enquiries. The DH will closely monitor the recall. Investigation is continuing.
Use of unregistered pCms may pose public health threats as their safety, efficacy and quality have not been proven. So far, the DH has not received reports of related adverse incidents.
"According to Section 119 of the Chinese Medicine Ordinance, Cap. 549, Laws of Hong Kong, no person shall sell, import or possess any pCm unless the pCm is registered. The maximum penalty involved is $100,000 and two years' imprisonment. On completion of the investigation, the DH will work with the Department of Justice for prosecution matters. The DH will also refer this case to the CMCHK for consideration of possible disciplinary action," the spokesman said.
The spokesman urged the public who have obtained the unregistered pCms to stop using them immediately and surrender the products to the Chinese Medicine Division of the DH at 16/F, AIA Kowloon Tower, Landmark East, 100 How Ming Street, Kwun Tong, Kowloon, during office hours. They should consult health-care professionals in case of doubt or if feeling unwell.
The list of registered pCms can be viewed at the following link: www.cmchk.org.hk/pcm/eng/#main_dis.htm .