Press Release
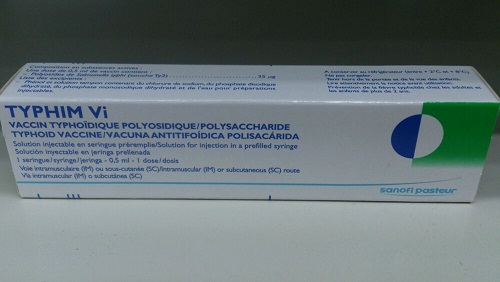
Total recall of Typhim VI (Typhoid Vaccine)
14 September 2012
The Department of Health (DH) today (September 14) instructed a licensed drug wholesaler, Sanofi-Aventis Hong Kong Limited (Sanofi), to conduct a total recall of Typhim VI (Typhoid Vaccine) (Registration number: HK-36827) from the market because of a quality issue.
A DH spokesman said that Sanofi informed DH today that according to the French manufacturer, Sanofi Pasteur S.A., several out-of-specification (OOS) test results regarding the antigen content were reported for Typhim VI, which might affect the efficacy of the vaccine. The root cause of the OOS test results is being investigated by the manufacturer.
Typhim VI had been supplied to DH clinics, private doctors, private hospitals, pharmacies and exported to Macau.
The DH has requested Sanofi to provide a detailed investigation report on the cause of the incident. The DH's investigation is continuing.
Typhim VI is indicated for the prevention of typhoid fever. It can only be sold on prescription and under the supervision of pharmacists at registered dispensaries.
The DH has not received any adverse report in connection with the product.
Sanofi has set up a hotline at 9316 7212 to answer related enquiries. The DH will closely monitor the recall.
"Members of the public who have been given the vaccine should consult their health-care providers for advice," the DH spokesman said.