Press Release
Warning on proprietary Chinese medicine containing undeclared Western drug ingredient
23 August 2012
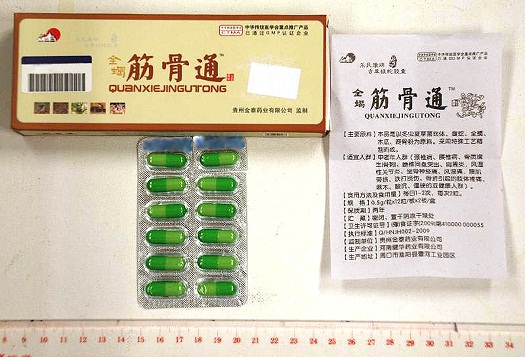
The Department of Health (DH) today (August 23) appealed to members of the public not to buy or consume an unregistered proprietary Chinese medicine (pCm) called "Quan Xie Jin Gu Tong", as it may contain undeclared Western drug ingredients that are dangerous to health.
The appeal followed the DH's receipt of notification from the Hospital Authority (HA) about a 68-year-old male patient. The DH commenced investigation immediately.
"The patient sought treatment from Pamela Youde Nethersole Eastern Hospital for peptic ulcer on August 14. He described a history of use of the above pCm and other western medicines for his muscle pain before. A drug-related adverse effect was suspected clinically. He was in a stable condition and was discharged on the next day.
"The HA's laboratory test on the pCm sample showed the presence of the undeclared Western medicine diclofenac, piroxicam and prednisone acetate. Investigation thus far reveals that the patient purchased the pCm from Huiyang in the Mainland. The pCm is not registered in Hong Kong. The DH investigation continues," a DH spokesman said.
"Diclofenac and piroxicam are non-steroidal anti-inflammatory drugs used to relieve pain and inflammation. The known side effects include gastro-intestinal discomfort, nausea and peptic ulcer. Prednisone acetate is a steroid and a prescription-only medicine. Taking prednisone acetate for a long time, especially in substantial dosages, can cause side effects such as moon face, high blood pressure, high blood sugar, muscle atrophy, peptic ulcer and even osteoporosis," the spokesman added.
The matter will be referred to our Mainland counterpart for their assessment and necessary follow-up.
"People must stop taking this pCm immediately and consult healthcare professionals as soon as possible if they feel unwell or are in doubt after taking the product. Members of the public are urged not to buy or consume products of unknown composition or from doubtful sources," the spokesman urged.