Press Release
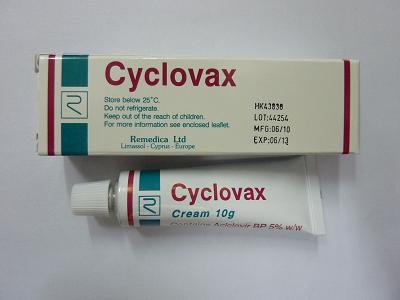
Recall of Cyclovax Cream 5%
4 May 2011
The Department of Health (DH) today (May 4) instructed the recall from consumers of a batch (Lot: 44254) of "Cyclovax Cream 5% " (registration no. HK-43836), a drug registered by Healthcare Pharmascience Ltd (Pharmascience), the product registration certificate holder, because of insufficient active ingredient in the product.
A spokesman explained that during routine market surveillance, samples of "Cyclovax Cream 5% " were purchased by DH inspectors and chemical analysis performed by the Government Laboratory revealed that only 3.2% instead of 5% of the active ingredient aciclovir was identified.
Initial investigation indicated that a total of 2 400 tubes of the affected batch of "Cyclovax Cream 5%", manufactured in Cyprus, had been imported by Pharmascience. The product had been sold to pharmacies and private doctors in Hong Kong and also exported to Macao.
"Cyclovax Cream 5% " is a topical antiviral drug for the treatment of herpes simplex virus skin infection. It requires doctors' prescription and can only be sold in dispensaries, under the supervision of pharmacists.
The spokesman said, "The lowered active ingredient content is a quality defect which can lead to ineffective treatment. Thus, healthcare professionals and retailers should stop supplying the said product to their clients. People who have used the affected product should consult healthcare providers if in doubt or feel unwell.
"Here, contravention of Hong Kong Law Cap 132, the Public Health and Municipal Services Ordinance, selling drug not of the nature, substance or quality demanded by the purchaser might have occurred. The maximum penalty involved is $10,000 and 3 months imprisonment. We will work with the Department of Justice for legal processing."
Pharmascience has set up a hotline 2565 6309 to handle related enquiries during office hours.
DH's will closely monitor the recall and also continue to investigate into the quality of other batches of the product.