Press Release
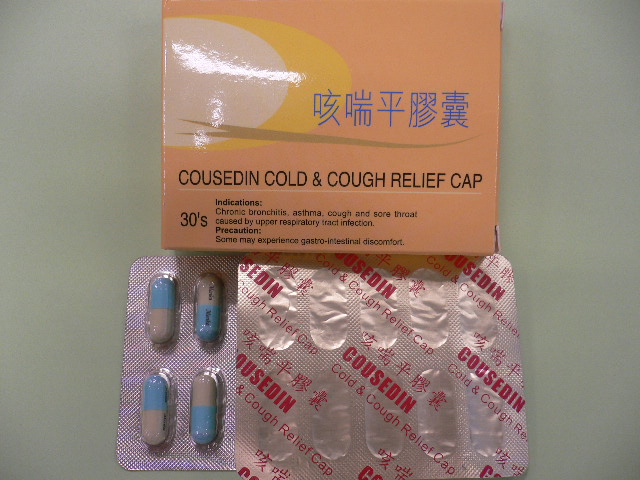
Recall of proprietary Chinese medicine "Cousedin Cold & Cough Relief Cap"
4 Nov 2010
The Department of Health (DH) today (November 4) urged members of the public not to buy or use a proprietary Chinese medicine (pCm) called "Cousedin Cold & Cough Relief Cap" as it was found to exceed the permitted microbial limit.
The product is manufactured by a local licensed pCm manufacturer, Merika Medicine Factory Limited, and is indicated for cough relief.
The matter began on October 29 when Macao Health Bureau (MHB) had detected excessive amounts of microorganisms in "Cousedin Cold & Cough Relief Cap" (batch number: 724345) during a surveillance exercise. The Bureau then instructed the local wholesaler to initiate a recall and also notified DH about the findings.
Upon receiving the notification, DH mounted an investigation immediately. It was revealed that the concerned batch was manufactured in 2008 and was for export to Macao only.
During inspection examination of the plant, DH obtained samples of different batches of the same product along with other products manufactured by the concerned manufacturer for laboratory analyses. Results available today showed that the only other batch of Cousedin Cold & Cough Relief Cap ever produced (batch no. 927111) also exceeded the microbial limit. So far, no report has been received about consumers feeling unwell after having taken the product.
Record indicated that there was a total of 2, 230 boxes in the above affected batch, of which 853 had been launched to the Hong Kong market only. While investigation will continue to ascertain the cause of contamination, DH has instructed the manufacturer to recall all of the Cousedin batch no. 927111 still in the local market. The manufacturer has already set up a hotline (2691 6631) to answer public enquiries about the recall.
Consumers may also choose to submit the recalled products to DH's Chinese Medicines Section at 2/F, Public Health Laboratory Centre, 382 Nam Cheong Street, Kowloon during office hours or destroy them before disposal.
A DH spokesman remarked that the MHB has also been notified. He reminded the public who has the product in hand to stop using it immediately and seek advice from healthcare professionals if they feel unwell.