Press Release
Products of Proprietary Chinese medicine with western drug ingredients found
5 August 2005
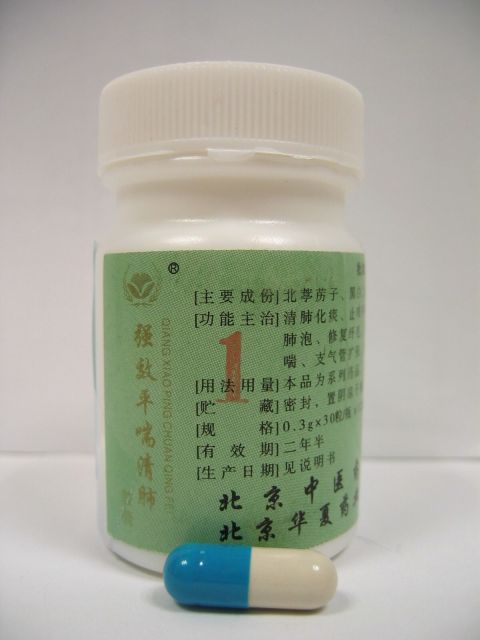
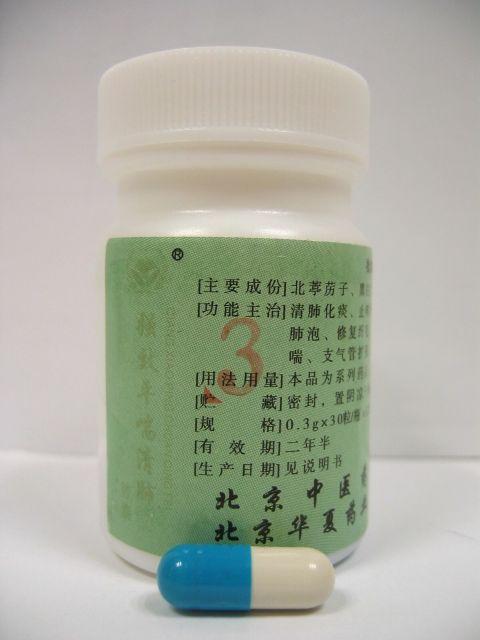
The Department of Health (DH) today (August 5) called on members of the public not to buy or use two products of a brand of proprietary Chinese medicine, namely Qiangxiao Pingchuan Qingfei Jiaonang(1) "????????(1)" and Qiangxiao Pingchuan Qingfei Jiaonang(3) "????????(3)" , both of which contain six western drug ingredients and may cause side effects.
Five of the six ingredients, morphine (??), codeine (???), theophylline (??), diclofenac (????), nifedipine (????) are prescription-only drugs and they should only be used under medical supervision. Cyproheptadine (???) can be bought over-the-counter.
Morphine and diclofenac are pain killers. Codeine is a cough suppressant. Theophylline is used to treat asthma. Nifedipine is used for hypertension. Cyproheptadine is used to relieve allergy.
Morphine and codeine can cause constipation, drowsiness and respiratory depression. Theophylline can cause irregular heart rate, gastro-intestinal discomfort and nausea. Diclofenac can cause gastro-intestinal discomfort. Nifedipine can cause headache and dizziness. Cyproheptadine can cause dizziness and drowsiness. Products containing the above named drug ingredients, except cyproheptadine, are classified as Third Schedule poisons under the Pharmacy and Poisons Ordinance, i.e. they can only be sold on a doctor's prescription and dispensed under a pharmacist's supervision. Products containing cyproheptadine and can be purchased over-the-counter.
The appeal was made after an investigation into a case in which a 68-year-old man admitted into United Christian Hospital on June 14 this year because of fever, shortness of breath and increased heart rate. The man claimed that he had taken the product bought from the Mainland for chronic obstructive pulmonary disease (COPD) for two months. He had recovered and was discharged home on June 29.
The DH has no record of these two products having been imported into Hong Kong for sale, or having been submitted for registration. The relevant authorities in the Mainland and Macao have been informed.
Members of the public who have been using the products are advised to immediately stop taking them and to seek medical attention if they feel unwell.
They are urged to dispose of the products or submit them to the DH's Pharmaceutical Service at the third floor of the Public Health Laboratory Centre at 382, Nam Cheong Street, Kowloon during office hours. People with COPD should consult medical professionals for appropriate advice and treatment.
DH has also set up a hotline 2319 2839 for enquiries during office hours.
People with COPD should consult medical professionals for appropriate advice and treatment.