Press Release
Warning on oral product containing banned Western drug ingredient
14 December 2012
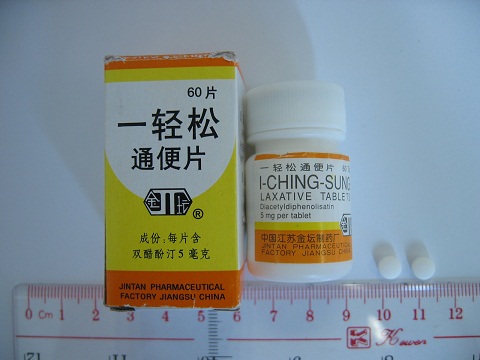
The Department of Health (DH) today (December 14) appealed to members of the public not to buy or consume an oral product called "Jin Tan 1-Ching-Sung Laxative Tablets", as it may contain a banned Western drug that is dangerous to health.
The appeal followed the DH's receipt of information from the Hospital Authority about a 92-year-old male patient who was admitted to Queen Mary Hospital for management of mild confusion. It was incidentally found that he had the history of using "Jin Tan 1-Ching-Sung Laxative Tablets" labelled as containing banned Western drug diacetyldiphenolisatin (also known as oxyphenisatin) for constipation for a few years. He is now in stable condition.
"According to the relative of the patient, the product was purchased from a local medicine shop in Central. The DH commenced investigation immediately and during the investigation, a 61 year-old male was arrested by the Police for sale of unregistered pharmaceutical product," a DH spokesman said.
DH's investigation is continuing.
According to the Pharmacy and Poisons Ordinance, sale of an unregistered pharmaceutical product is a criminal offence and the maximum penalty for this offence is a fine of $100,000 and two years' imprisonment.
"Diacetyldiphenolisatin was banned for its hepatotoxicity in Hong Kong in 1997. It was used previously for treating constipation," the spokesman explained.
The spokesman urged members of the public to stop taking the product immediately if they still have the product in their possession. People should consult healthcare professionals if they feel unwell or are in doubt after taking the product.
"They should submit it to the DH's Drug Office at Room 1801, 18/F, Wu Chung House, 213 Queen's Road East, Wan Chai, Hong Kong, during office hours as soon as possible because it is an unregistered pharmaceutical product containing a banned drug ingredient," the spokesman continued.
"Patients with constipation and any chronic disease ought to consult healthcare professionals for appropriate advice and management. They are strongly urged to refrain from self-medication or using over-the-counter products without professional supervision," the spokesman concluded.